Updated May 16, 2026
Exploring synthesis possibilities of complex structures
Computational chemist: run multiple parameter regimes to surface classical and novel disconnections.
For complex targets where the "right" disconnection isn't obvious, running the model under multiple parameter regimes surfaces both the classical strategy and the unconventional ones. Use this when you'd rather brainstorm than commit.
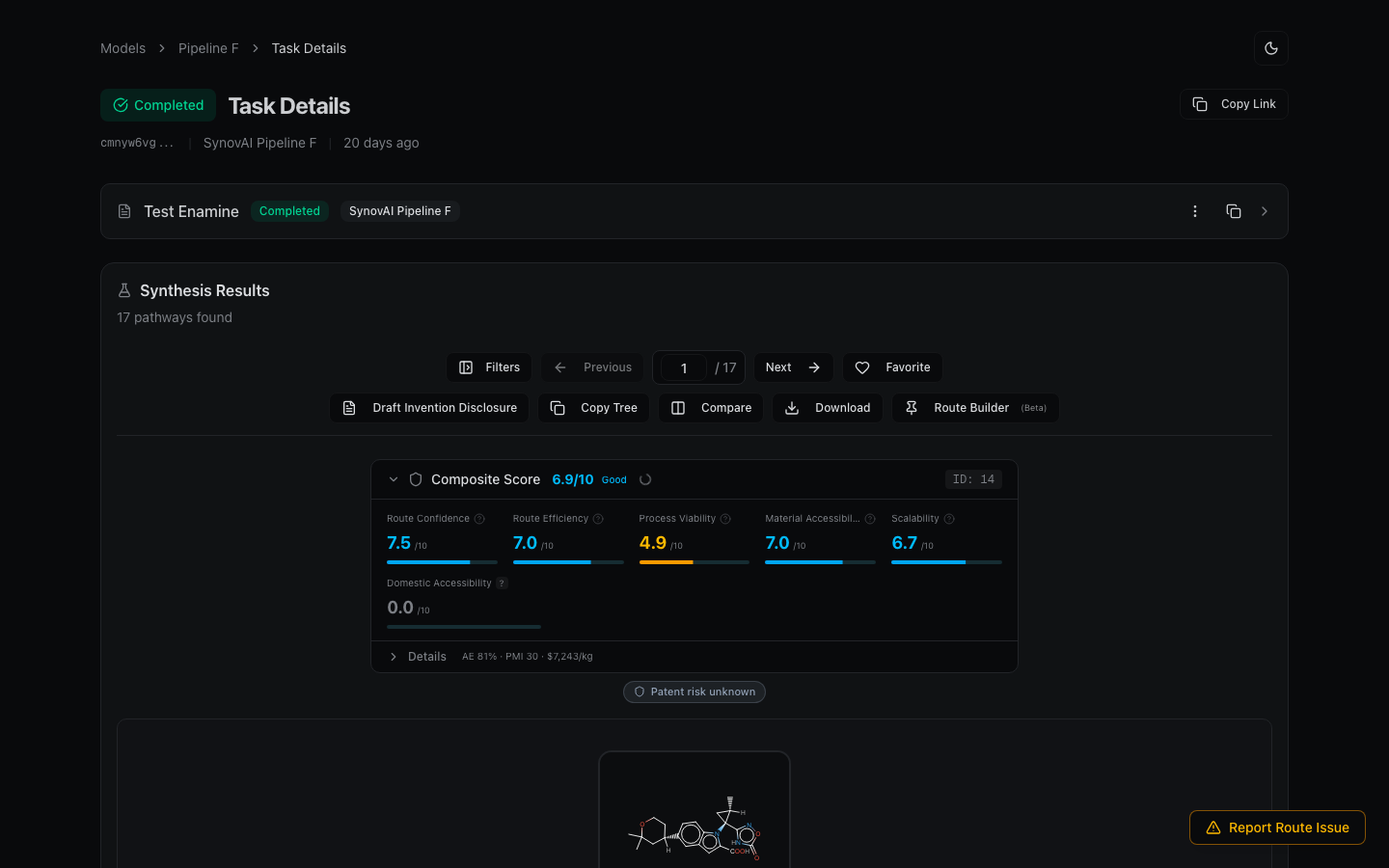
Steps
- Submit your target with a conservative parameter set first: moderate route length (4-6 steps), Standard precedent, deep search on. This produces the model's best classical strategy.
- Wait for the result, then resubmit the same target with Novel precedent. This pushes the model toward unconventional disconnections it would normally rank lower.
- (Optional) Run a third pass with a longer route length (8) and Exploratory precedent. Even slower, but surfaces routes the tighter searches won't.
- Compare results across all submissions in the Recent Tasks table. Use the task search (Structure mode) to filter to runs of the same target.
- Study the disconnections that show up in the Novel runs but not the conservative ones. These are usually where the model is suggesting bond formations that aren't in the textbook for this class. Some will be wrong; some will be genuinely creative.
- Pin the best step from each run into Route Builder to assemble a hybrid route across runs.
Output
A multi-perspective view of how to make the target. Not all routes are equally credible, but the breadth gives you something the single-best-answer approach can't: a sense of the disconnection space the target sits in.